Research
What is ‘tau’ and what role does it play in dementia?
In the laboratory, dementia researchers focus on understanding how sickness in the brain can lead to loss of memory and identity. Over many studies, a few key proteins have stood out as having a significant role, and one of them is tau.
Dr Miguel Ramirez Moreno studies tau through his work as a postdoctoral researcher with Professor Amrit Mudher, through her AS funded project grant. Miguel has put together this helpful explainer on tau's function in dementia.
What is tau?
Tau is a protein naturally produced by our cells, helping them function properly. Its roles include supporting the internal structure of cells - by acting as a kind of strengthening scaffolding - to make them stronger.
This is especially important for our neuron cells, as they need to be made up of strong and resilient parts, to communicate with each other, transporting everything from electric signals to vital nutrients
Good maintenance by proteins like tau keeps our bodies working efficiently. However, tau has gained a negative reputation because it is involved in several diseases that lead to dementia, including the most common one, Alzheimer’s disease.
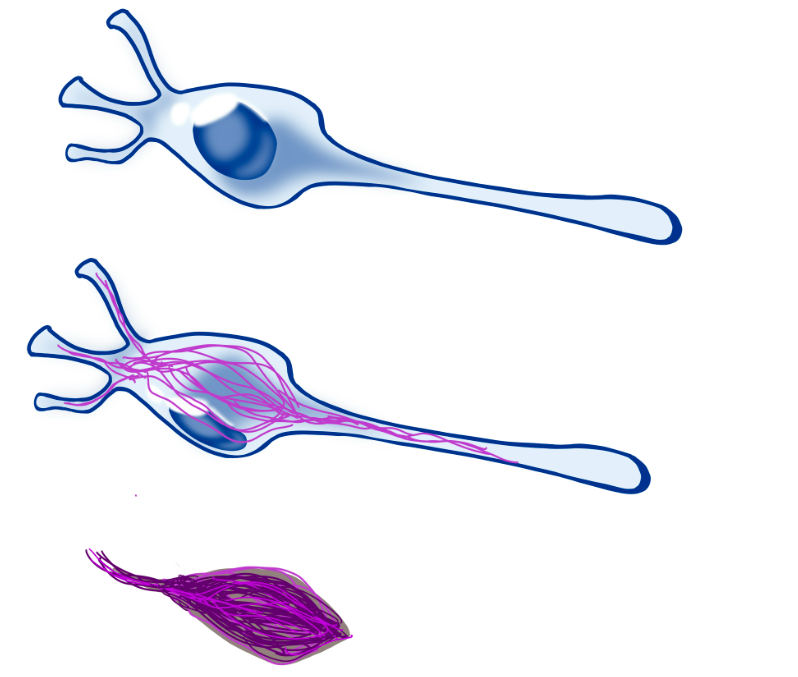
In the image above, we see a healthy neuron (top). tau acts normally across the structure of a neuron, but abnormal tau starts forming fibrous tangles (centre), that eventually are all what remains of the neuron (bottom).
Did you know... We all have tau inside our brains – inside every one of our cells. And normally that is fine. The problem is not the protein itself, but its behaviour.
Untangling findings on tau and Alzheimer’s disease
Dementia is not a single disease, but a syndrome caused by many different conditions affecting the brain. Alzheimer’s disease is responsible for more than half of all the diagnosed cases of dementia.
The disease was first spotted over a century ago, by Alois Alzheimer. He described the strange clumps he’d seen inside of the brains of people affected - fibre-like structures, or “tangles”.
As is often the case with major scientific breakthroughs, it took a collaborative effort to understand what these fibres actually were.
In 1975, a group at Princeton University (USA) discovered tau as a protein involved in protecting the internal structure of the cells. But it was not until 1988 that several scientists at Cambridge University (UK) found out that tau was making the tangles that Alzheimer described decades earlier.
The mystery of the tangles Alois Alzheimer had seen was solved: Finding tau was present in a form it should not be in Alzheimer’s disease. But this opened up more questions for researchers of Alzheimer’s disease. what Was this just an unusual presentation or something more? Could tau be responsible of for damaging our neurons getting hurt?
How relevant is tau for dementia?
Alzheimer’s disease is marked by the presence of abnormal tau tangles, alongside excessive depositions of amyloid beta – commonly known as senile plaques.
Scientists have discovered many ways in which these two proteins interact in the disease process. According to the amyloid cascade hypothesis, amyloid beta is disrupted first, which subsequently leads to changes in tau and other proteins. Abnormal tau is also linked to many other forms of dementia, either on its own or in combination with other proteins. This is why these conditions are collectively referred to as ‘tauopathies’. In each of these diseases, tau may be more or less important, but it is always involved.
What do and don’t we know about tau?
When tau protein becomes abnormal, it stops doing its usual job of keeping brain cells healthy. This leaves the cells more vulnerable to damage. But it doesn’t just stop helping, it can start harming the brain.
Scientists have found that abnormal tau can move from one neuron to another, which helps explain how the damage spreads across the brain in diseases like Alzheimer’s.
What research are Alzheimer's Society supporting into tau?
Research will beat dementia and will lead to improved diagnosis, effective treatments and the high-quality care that everyone living with dementia deserves. Learn more about some of the research we are funding to answer the outstanding questions about the tau protein, and how it could be targeted by future treatments.


Normally, the body clears out unnecessary tau, but in people with diseases like Alzheimer’s disease, it builds up and harms brain cells eventually causing the cells to die and the symptoms of dementia appear. Dr Croft’s previous research shows that tau knots can unravel on their own, but very slowly.
Dr Croft’s project will help us to understand how tau breaks down and whether we can speed up this process to prevent harmful tangles from forming. By identifying ways to boost tau removal, researchers could develop new treatments that could slow or prevent Alzheimer’s disease.
Find out more about this research project
Researchers know the tau protein can move from one nerve cell to another through the areas where two cells connect. This is how the disease spreads through the brain in Alzheimer’s disease, making the symptoms worse over time.
Professor Mudher is using clever genetic manipulation in fruit flies to turn genes on and off in their nerve cells. This will reveal the genes responsible for helping tau spread between brain cells.
Find out more about this research project
During sleep, the brain’s natural cleaning system called the glymphatic system flushes out waste. Dr Harrison’s previous research has shown that tau can also be cleared away by the glymphatic system.
However, as people age, this cleaning system becomes less effective. Dr Harrison and his team will investigate whether a new drug can boost the brain’s waste removal process, helping to slow or prevent memory loss.
Find out more about this research project